Authors: Chirag H. Patel, Yi Dong, Navid Koleini, Xiaoxu Wang, Brittany L. Dunkerly-Eyring, Jiayu Wen, Mark J. Ranek, Laura M. Bartle, Daniel B. Henderson, Jason G. Sagert, David A. Kass, and Jonathan D. Powell
Brief Summary: Phosphorylation at Serine-1365 on TSC2 dampens T-cell activation response. Preventing this generates more proliferative T cells that survive adverse environments and enhance tumor adoptive cell therapy.
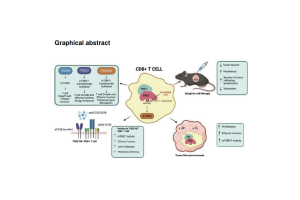
Abstract: MTORC1 integrates signaling from the immune microenvironment to regulate T cell activation, differentiation, and function. TSC2 in the tuberous sclerosis complex tightly regulates mTORC1 activation. CD8+ T cells lacking TSC2 have constitutively enhanced mTORC1 activity and generate robust effector T cells; however sustained mTORC1 activation prevents generation of long-lived memory CD8+ T cells.
Here we show manipulating TSC2 at Ser1365 potently regulates activated but not basal mTORC1 signaling in CD8+ T cells. Unlike non-stimulated TSC2 knockout cells, CD8+ T cells expressing a phospho-silencing mutant TSC2-S1365A (SA) retain normal basal mTORC1 activity. PKC and T-cell Receptor (TCR) stimulation induces TSC2 S1365 phosphorylation and preventing this with the SA mutation markedly increases mTORC1 activation and T-cell effector function. Consequently, SA CD8+ T cells display greater effector responses while retaining their capacity to become long-lived memory T cells. SA CD8+ T cells also display enhanced effector function under hypoxic and acidic conditions.
In murine and human solid-tumor models, CD8+ SA T cells used as adoptive cell therapy display greater anti-tumor immunity than WT CD8+ T cells. These findings reveal an upstream mechanism to regulate mTORC1 activity in T cells. The TSC2-SA mutation enhances both T cell effector function and long-term persistence/memory formation, supporting an approach to engineer better CAR-T cells for treating cancer.